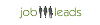Salário médio: €350 /mensal
Mais estatísticasA Randstad Professionals é a unidade de negócio do Grupo Randstad, direccionada para o recrutamento de Middle & Top Management.
O nosso cliente é um prestigiado Laboratório de Serviços Farmacêuticos, que pretende integrar um Regulatory Affairs Specialist.
...of the results sent by the external vendor);
- Monitoring of regulatory submissions PV related;
- Submission of PV clinical... ...- Audits support;
- Customer Service Support;
Regulatory Affairs:
- Support MS submissions (DCP Submissions, variations within...
...In the context of our growing activities, we are looking for a Regulatory Affairs Specialist to join our teams in Portugal.
You will integrate a team of Regulatory Affairs professionals dedicated to Southern Europe and will have the mission to develop PLG’s local Portugal...
...around for a new challenge?
Test the waters and find out if you are our new RA Manager.
GENERAL INFORMATION:
Position: Regulatory Affairs Manager – CMC (m/f/x)
Location: one of our offices in Porto, Heidenheim an der Brenz, Berlin, or remote (Portugal/Germany)...
...Regional Regulatory Affairs Senior Associate page is loaded Regional Regulatory Affairs Senior Associate
Apply locations Portugal - Lisbon time type Full time posted on Posted 4 Days Ago job requisition id R-186480 Career Category
Regulatory Job Description...
...accelerate customer success. We translate unique clinical, medical affairs and commercial insights into outcomes to address modern market... ...quality standards are maintained.
• Adheres to established regulatory standards, including but not limited to ICH-E3 guidelines, as...
Trabalho remoto
...A empresa onde vai trabalhar
Integre uma empresa multinacional farmacêutica de referência assumindo a função de Medical Affairs Manager.
A sua nova função
Este profissional será o responsável pela definição da estratégia médica para os produtos de diferentes...
Descrição do emprego
Estamos à procura de um Analista para o nosso departamento de Crédito e Relatórios Regulatórios. Esta posição envolve o cálculo e análise de Ativos Ponderados pelo Risco (RWA) e a preparação de relatórios regulatórios como COREP, divulgações do...
...com presença em Portugal e internacional e está a recrutar um Regulatory Reporting Senior (m/f/d)
A sua nova função
Integrado no... ...responsabilidades:
Preparação atempada, precisa e eficiente, de reportes regulatórios às entidades de supervisão;
Avaliação do cumprimento dos...
Expleo is a trusted partner for your innovation journey. As a global engineering, technology and consulting service provider, we are ideally positioned to help you achieve your ambitions and future-proof your business. With a smart blend of bold thinking and reliable execution...
...and overseeing the medical strategy, ensuring compliance with regulatory requirements, and fostering relationships with healthcare professionals... ...Requirements
~3-5 years of previous experience as a Medical Affairs Advisor in a pharmaceutical company
~ Previous experience as...
...worked to support the passage of numerous bills that have benefitted close to 500 million people in developing countries.
Political Affairs Internship: The role focuses on making poverty a focus of U.S., Canadian and U.K. foreign policy. This is a part-time 12-hours per...
Trabalho remoto
...you want to have a meaningful impact on the financial services industry Do you see the opportunity to improve legal compliance and regulatory processes with intelligent technology If so at Apiax we are enabling solutions for global financial institutions through embedded...
...Description
Site Start-Up Regulatory Coordinator with Portuguese AND/OR Italian / single sponsor (FSP)/Site Contracts support... ...accelerate customer success. We translate unique clinical, medical affairs and commercial insights into outcomes to address modern market...
...meaningful impact on the financial services industry If so then we have a great opportunity for you.
Were looking for a Junior Regulatory Engineer to join our RegTech company.
You are a legal & compliance professional who is technologically adept or a...
...cumprimento com os requisitos legais em vigor.
Responsabilidades :
Preparação atempada, precisa e eficiente, de reportes regulatórios às entidades de supervisão;
Avaliação do cumprimento dos requisitos legais em vigor;
Implementação de controlos de...
...accelerate customer success. We translate unique clinical, medical affairs and commercial insights into outcomes to address modern market... ...where everyone feels like they belong.
JOB SUMMARY
The Regulatory Intelligence Department at Syneos Health impact decision-...
...Regulatory Compliance Manager - Portugal
Regulatory Compliance Manager - Portugal
Remote: Portugal About Revolut
People deserve more from their money. More visibility, more control, more freedom. And since 2015, Revolut has been on a mission to deliver just...
...Job Description - Manager Regulatory Medical Writing, Oncology (2406179316W)
Manager Regulatory Medical Writing, Oncology - 2406179316W
Description
Manager Regulatory Medical Writing, Oncology
At Johnson & Johnson Innovative Medicine, what matters most is...
...Senior Public Affairs Consultant – Lisbon
What profile are we looking for?
This role requires a versatile and flexible person,... ...strategic partners to navigate the current political, legislative, regulatory, and social complexity.
This is a unique opportunity for a...
...Coordinating all committees that ensure continuous adherence to the laws and notably in charge of:
Participating to the design of any regulatory-related 1st level controls plan
Interacting with all relevant partners & functions to identify potential area of concern and...
...difference in the field of Diabetes? Do you have experience in Medical Affairs and a strong background in Diabetes or Cardiovascular Disease?... ...team, which is integrated into our Clinical, Medical, and Regulatory (CMR) department. The CMR department at Novo Nordisk is at the...
...they are conducted in accordance with established protocols, regulatory guidelines, and company standards. You will be a key player in... ...investment in Portugal, when comparing 2020 with 2023.
In Medical Affairs, we drive business with science and we partner for better care...
...mindset
About the department
In the Clinical Medical and Regulatory (CMR) department we drive change to defeat serious chronic... ...investment in Portugal, when comparing 2020 with 2023.
In Medical Affairs, we drive business with science and we partner for better care...
...equivalent degree in a scientific area; A candidate with experience in a Quality Assurance related position; A candidate with Regulatory Affairs basic knowledge and Quality aspects namely cGMP, ICH guidelines and GDP; A proactive candidate with ability to lead complex...
...with staff based across Europe, North America and Asia all covering services within: Medical Information, Pharmacovigilance, Regulatory Affairs and Quality Assurance.
PrimeVigilance provides first class support to our small to large pharmaceutical and biotechnology...
...product launches and services for distribution partners
acting as an interface between our client and partners such as suppliers, regulatory affairs, legal and marketing
leading, developing and controlling distribution partners including regular business reviews, changes...
...professional will ensure the implementation of the oncology medical affairs strategy to support the business plan and conduct all relevant... ...preferred.
Excellent knowledge and understanding on the regulatory guidelines and fluency in English is required.
What you'...
...accelerate customer success. We translate unique clinical, medical affairs and commercial insights into outcomes to address modern market... ...projects (e.g. submissions, integrated analyses), and attend regulatory agency meetings or responds to questions, as needed, to...
Trabalho remoto
...accelerate customer success. We translate unique clinical, medical affairs and commercial insights into outcomes to address modern market... ...deliverables with a focus on patient safety, protocol/GCP/regulatory compliance and data integrity.
Oversees site interactions...
Trabalho remoto